Summary: Complement C3, a chemically modified type of an inflammatory immune protein, was extra plentiful in autopsy mind tissue of ladies who died from Alzheimer’s illness than in males.
Source: Scripps Research Institute
Scientists at Scripps Research and Massachusetts Institute of Technology (MIT) have discovered a clue to the molecular reason behind Alzheimer’s—a clue that will additionally clarify why girls are at larger danger for the illness.
In the research, reported on December 14, 2022, in Science Advances, the researchers discovered {that a} notably dangerous, chemically modified type of an inflammatory immune protein referred to as complement C3 was current at a lot greater ranges within the brains of ladies who had died with the illness, in comparison with males who had died with the illness.
They additionally confirmed that estrogen—which drops in manufacturing throughout menopause—usually protects in opposition to the creation of this type of complement C3.
“Our new findings suggest that chemical modification of a component of the complement system helps drive Alzheimer’s, and may explain, at least in part, why the disease predominantly affects women,” says research senior creator Stuart Lipton, MD, PhD, professor and Step Family Foundation Endowed Chair within the Department of Molecular Medicine at Scripps Research and a medical neurologist in La Jolla, California.
The research was a collaboration with a staff led by Steven Tannenbaum, PhD, Post Tenure Underwood-Prescott Professor of Biological Engineering, Chemistry and Toxicology at MIT.
Alzheimer’s, the most typical type of dementia that happens with getting older, at present afflicts about six million individuals within the U.S. alone. It is all the time deadly, normally inside a decade of onset, and there’s no authorized remedy that may halt the illness course of, not to mention reverse it.
The shortcomings of therapies replicate the truth that scientists have by no means totally understood how Alzheimer’s develops. Scientists additionally don’t know totally why girls account for almost two-thirds of instances.
Lipton’s lab research biochemical and molecular occasions that will underlie neurodegenerative ailments, together with the chemical response that types a modified kind of complement C3—a course of referred to as protein S-nitrosylation.
Lipton and his colleagues beforehand found this chemical response, which occurs when a nitric oxide (NO)-related molecule binds tightly to a sulfur atom (S) on a selected amino acid building-block of proteins to type a modified “SNO-protein”.
Protein modifications by small clusters of atoms reminiscent of NO are widespread in cells and usually activate or deactivate a goal protein’s capabilities. For technical causes, S-nitrosylation has been tougher to review than different protein modifications, however Lipton suspects that “SNO-storms” of those proteins could possibly be a key contributor to Alzheimer’s and different neurodegenerative issues.
For the brand new research, the researchers used novel strategies for detecting S-nitrosylation to quantify proteins modified in 40 postmortem human brains. Half of the brains have been from individuals who had died of Alzheimer’s, and half have been from individuals who hadn’t—and every group was divided equally between men and women.
In these brains, the scientists discovered 1,449 completely different proteins that had been S-nitrosylated. Among the proteins most frequently modified on this manner, there have been a number of which have already been tied to Alzheimer’s, together with complement C3. Strikingly, the degrees of S-nitrosylated C3 (SNO-C3) have been greater than six-fold greater in feminine Alzheimer’s brains in comparison with male Alzheimer’s brains.
The complement system is an evolutionarily older a part of the human immune system. It consists of a household of proteins, together with C3, that may activate each other to drive irritation in what is named the “complement cascade.”
Scientists have identified for greater than 30 years that Alzheimer’s brains have greater ranges of complement proteins and different markers of irritation, in comparison with neurologically regular brains. More latest analysis has proven particularly that complement proteins can set off brain-resident immune cells referred to as microglia to destroy synapses—the connection factors by way of which neurons ship indicators to at least one one other.
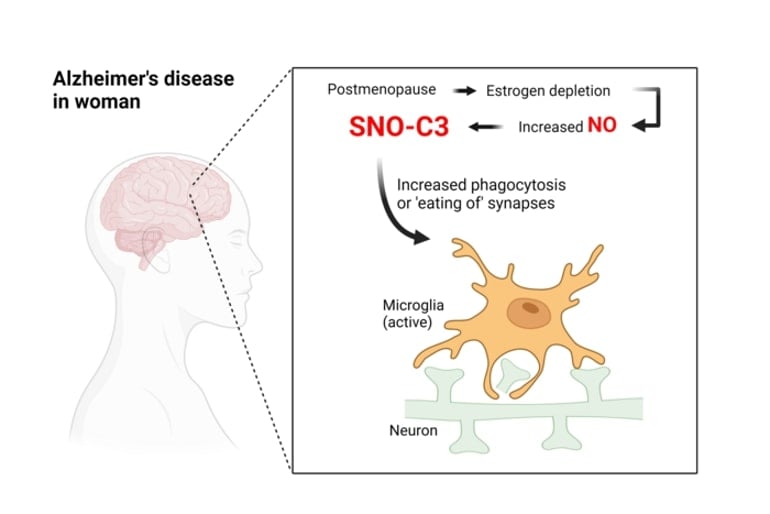
Many researchers now suspect that this synapse-destroying mechanism a minimum of partly underlies the Alzheimer’s illness course of, and lack of synapses has been demonstrated to be a major correlate of cognitive decline in Alzheimer’s brains.
Why would SNO-C3 be extra widespread in feminine brains with Alzheimer’s? There has lengthy been proof that the feminine hormone estrogen can have brain-protective results beneath some situations; thus, the researchers hypothesized that estrogen particularly protects girls’s brains from C3 S-nitrosylation—and this safety is misplaced when estrogen ranges fall sharply with menopause.
Experiments with cultured human mind cells supported this speculation, revealing that SNO-C3 will increase as estrogen (β-estradiol) ranges fall, because of the activation of an enzyme that makes NO in mind cells. This enhance in SNO-C3 prompts microglial destruction of synapses.
“Why women are more likely to get Alzheimer’s has long been a mystery, but I think our results represent an important piece of the puzzle that mechanistically explains the increased vulnerability of women as they age,” Lipton says.
He and his colleagues now hope to conduct additional experiments with de-nitrosylating compounds—which take away the SNO modification—to see if they’ll cut back pathology in animal fashions of Alzheimer’s and finally in people.
“Mechanistic insight into female predominance in Alzheimer’s disease based on aberrant protein S-nitrosylation of C3,” was co-authored by Hongmei Yang, Haitham Amal, John Wishnok, Sarah Lewis, and Steven Tannenbaum of the Massachusetts Institute of Technology; and Chang-ki Oh, Emily Schahrer, Dorit Trudler, Tomohiro Nakamura, and Stuart Lipton, of Scripps Research.
Funding: This research was supported partly by the National Institutes of Health (R01AG056259, R01 AG061845, R61 NS122098, RF1918 NS123298, R35 AG071734, RF1 AG057409, R01 AG056259, R56 AG065372, R01 919 DA048882, and DP1 DA041722).
About this Alzheimer’s illness analysis information
Author: Press Office
Source: Scripps Research Institute
Contact: Press Office – Scripps Research Institute
Original Research: The picture is credited to Chang-ki Oh and Stuart Lipton, Scripps Research
Original Research: Open entry.
“Mechanistic insight into female predominance in Alzheimer’s disease based on aberrant protein S-nitrosylation of C3” by Stuart Lipton et al. Science Advances
Abstract
Mechanistic perception into feminine predominance in Alzheimer’s illness primarily based on aberrant protein S-nitrosylation of C3
Protein S-nitros(yl)ation (SNO) is a posttranslational modification concerned in various processes in well being and illness and might contribute to synaptic harm in Alzheimer’s illness (AD). To determine SNO proteins in AD brains, we used triaryl phosphine (SNOTRAP) mixed with mass spectrometry (MS).
We detected 1449 SNO proteins with 2809 SNO websites, representing a variety of S-nitrosylated proteins in 40 postmortem AD and non-AD human brains from sufferers of each sexes. Integrative protein rating revealed the highest 10 elevated SNO proteins, together with complement element 3 (C3), p62 (SQSTM1), and phospholipase D3. Increased ranges of S-nitrosylated C3 have been current in feminine over male AD brains.
Mechanistically, we present that formation of SNO-C3 relies on falling β-estradiol ranges, resulting in elevated synaptic phagocytosis and thus synapse loss and consequent cognitive decline. Collectively, we reveal strong alterations within the S-nitrosoproteome that contribute to AD pathogenesis in a sex-dependent method.






Discussion about this post